Home > Product Design & Engineering Services > User-Centered Design > Usability
Explore our other User-Centered Design services
How Contextual Inquiry dictates the design process
The best way to gain a deep understanding of end users and usability challenges is to observe them in the actual environment. Nectar utilizes contextual inquiries as a crucial part of the design process by observing users in context, which enables designers to understand and empathize with their needs. This process can take place in an operating room or in the comfort of a home depending upon the device. Information gathered directly feeds into the design requirements and user needs. We are experts at identifying even the smallest details, and asking the right questions that lead to outstanding user experiences.
Generating powerful insights with
Critical Task Analysis
We take risk assessment to the next level by conducting a critical task analysis. Simply put, this involves analyzing the results of the contextual inquiries, documenting workflows, and generating a known use related risk assessment to identify critical tasks. Critical tasks identify crucial moments in the use of the device in which serious harm or injury can occur to the user or patient if the task is preformed incorrectly or not at all. This step in the medical design process can be the difference between life and death. Nectar is dedicated to creating devices that are easy to use, but above all else safe and effective
We specialize in medical device Risk Analysis, Assessment and Management
Medical devices can make the difference between life and death. If a device is used frequently on many different patients in a hospital setting, the risk of causing harm to a patient or healthcare practitioner is elevated. For this reason, a lot of the medical device development process revolves around risk reduction.
Risk reduction is an important tool that involves the identification of potential hazards, as well as understanding how these hazards can be mitigated for users. Thorough analysis is required to enable designers to identify any possible flaws associated with the design – in both normal and fault conditions.
We are ISO 13485 Compliant through every step of the way


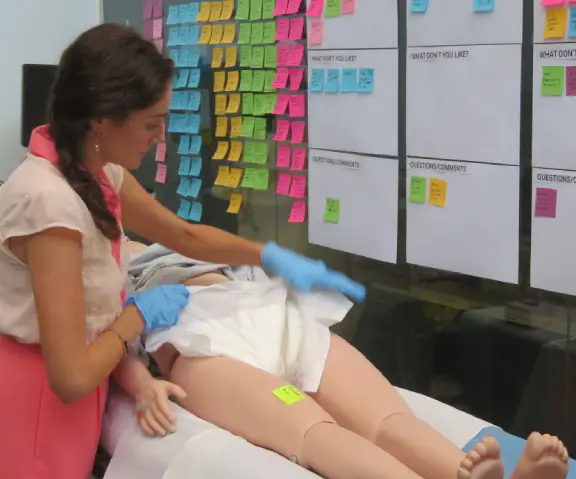

Formative Usability Testing in medical device development
Studies are an essential part of evaluating usability – they enable us to understand how a product is performing and how well-received it is by users. Here at Nectar we are big on formative studies as they help us identify and resolve usability issues, enabling us to make the design process as smooth as possible.
Used during the early stages of the design process, formative studies analyze the user experience (UX) to identify any potential issues early in the design process. Formative studies guide the rest of the design process, as they enable you to filter out different design features until only the ones that will benefit the end-users remain. As they are formative, these studies combine to move the iterative process forward; as the team discovers more potential issues, they can collate these findings to guide further development.

Summative Usability Testing in medical device development
Summative evaluation is specified by the International Electrotechnical Commission (IEC) 62366 for the evaluation of the usability of medical devices. In a nutshell, it is a methodology that ensures designers implement a user-first perspective. Once the design for a medical device has been concluded, summative testing is conducted to validate the safety of the product.
Not only are we at Nectar experts in summative evaluation, we can also do it well under pressure. The Covid-19 pandemic resulted in a desperate need for ventilators. We worked with BreathDirect to create the BDR-19™ Critical-Care Ventilator – an easy to use, mass produced, full featured critical care ventilator that was created in 10 months. In less than a year, we conducted summative evaluation studies to ensure the ventilator met the high bar for performance, quality, and safety.

Darren is an accomplished, dynamic leader who founded Nectar, X-Naut, and BreathDirect. He is committed to improving the world through the synthesis of technology, science, and art. Under Darren’s leadership, Nectar is leveraging the latest advances in cutting-edge technology to transform medical device development and design. As part of his

Dr. Steve Wells holds a critical role in steering Nectar’s operations and defining its strategic direction. His impactful leadership has established a clear course for the company’s success. He has revitalized the company with a passion for maximizing impact through expert resource utilization since taking the helm in 2020. Steve has boosted Nectar’s portfolio, streamlined processes, and reinvigorated its innovation agenda, all with the goal of developing life-saving and impactful products. His background in senior leadership roles at Georg Fischer and his people-focused, results-driven leadership style make him an invaluable asset to our team. Steve’s extensive knowledge in chemistry, biocompatibility, and biology only adds to his value as a team member.





Adam Marten has worked in the aerospace and consumer products industries since 2006 with lead engineering experience in conceptual design, product development, analysis and performance qualification testing. Adam has experience leading a range of technical projects, including multiple structural analyses for military and aerospace applications. He has also worked directly with engineering teams to develop medical devices and laboratory instrumentation.






With over two decades of experience in healthcare innovation, Kerry has been instrumental in transforming patient care and improving patient outcomes across the globe. Her extensive knowledge and expertise in the cardiovascular and cardiothoracic operating room, as well as her experience in emergency medicine, geriatrics, pediatric psychiatry, and main OR, make her a versatile and valuable member of the team. From small hospitals to large teaching institutions, Kerry’s 20 years of experience with patient documentation, including the use of EPIC, Cerner, and Meditech electronic charting systems, have positioned her as a leader in her field. Her recent involvement in helping transition the Providence St. Vincent Medical Center’s Cardiac Surgery program from paper charting to electronic documentation further underscores her commitment to advancing the healthcare industry.








