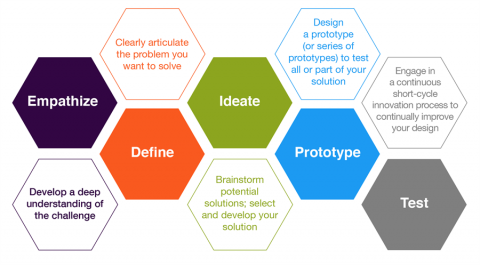
User testing is an essential method in the user-centered design process for medical devices. It involves engaging users in the evaluation and validation of design concepts and prototypes to gather feedback and insights. Here are some common methods used to test medical device designs and involve users in the process:
Contextual Inquiry
This method involves observing and interviewing users in their real-world environment to understand their needs, behaviors, and workflows. It helps designers gain insights into user requirements and identify potential design improvements.
Usability Testing
Usability testing focuses on evaluating the usability and user experience of a device by observing users as they interact with prototypes or working models. It helps identify any usability issues, user errors, or areas for improvement.
Focus Groups

Surveys and Questionnaires
Surveys and questionnaires can be used to gather feedback from a larger group of users. They can provide quantitative data on user preferences, satisfaction levels, and other relevant metrics.
Cognitive Walkthroughs
In a cognitive walkthrough, designers and users go through the design together, step-by-step, while the user explains their thoughts, assumptions, and decision-making process. This method helps identify potential usability issues and areas where users may struggle to understand or complete tasks.
Expert Reviews
In addition to involving users, it can be helpful to have domain experts, such as medical professionals or human factors engineers, review the design. Their expertise and knowledge can provide valuable insights and recommendations for improving usability and safety.
Iterative Design
User testing should be an iterative process, meaning that feedback from each round of testing is used to refine the design and create new prototypes. This iterative approach ensures that the final product meets user needs and expectations effectively.
These methods should be tailored to the specific context and requirements of the medical device being developed. It’s important to involve users throughout the design process to ensure that the device is intuitive, safe, and meets their needs effectively.
“The Product Development Playbook” offers a wealth of valuable information and practical insights for anyone involved in the field of product design. Whether you’re a seasoned professional or just starting your journey, this podcast provides a valuable resource to enhance your knowledge and skills. To listen to this episode in full – click here!