Navigating Regulatory Compliance and Design Controls in Medical Device Labeling: A Critical Exploration
In the realm of medical device manufacturing, adhering to regulatory standards such as the FDA’s guidelines, ISO 13485, and 21 CFR Part 801 is not just a matter of compliance but a critical step in ensuring patient safety and product effectiveness. Emphasizing design controls in medical device labeling, which includes every detail from label size to the placement of safety information, is pivotal in communicating vital information to users. This blog delves into the intricate aspects of medical device labeling, underscoring the importance of FDA design controls, symbols, instructions, warning labels, and more in the early stage product development process. For comprehensive insights into the design review and design control medical device strategies, medical product development process, visit Nectar.
The Essence of FDA-Regulated Medical Device Labeling
The FDA’s stringent regulations on medical device labeling are a cornerstone of ensuring safety and efficacy. Understanding these guidelines, including the specific requirements outlined in 21 CFR Part 801, is crucial for manufacturers. This section explores the mandatory labeling elements such as device identification, intended use, manufacturer identification, and warnings, which are essential for compliance and patient safety under medical device regulations.
Integrating Symbols and Instructions for Enhanced Clarity
The use of standardized symbols and concise instructions in medical device labeling is not only a regulatory requirement but also a best practice in risk management within the industry. Symbols provide a universal language that transcends linguistic barriers, ensuring that crucial control information and warnings are universally understood. This section discusses how effective symbol usage and clear instructions can significantly mitigate risks associated with medical device usage.
Label Size, Placement, and Printing Methods
The physical aspects of medical device labeling, such as label size, placement, and printing methods, play a pivotal role in ensuring that the product information is accessible and legible. This section covers the importance of these aspects in the product development design process, including material consideration and the choice of sustainable, durable printing methods. For more insights into medical product development, visit Nectar Product Development.
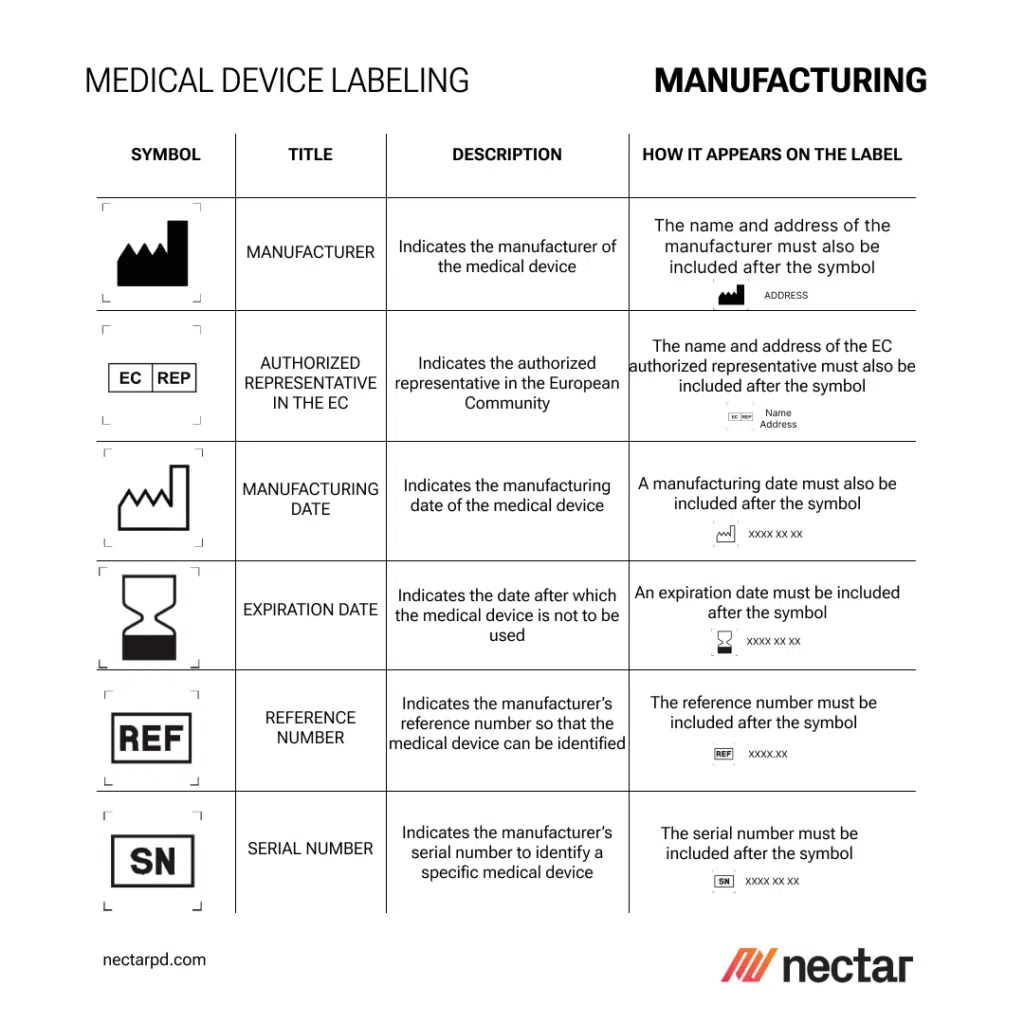
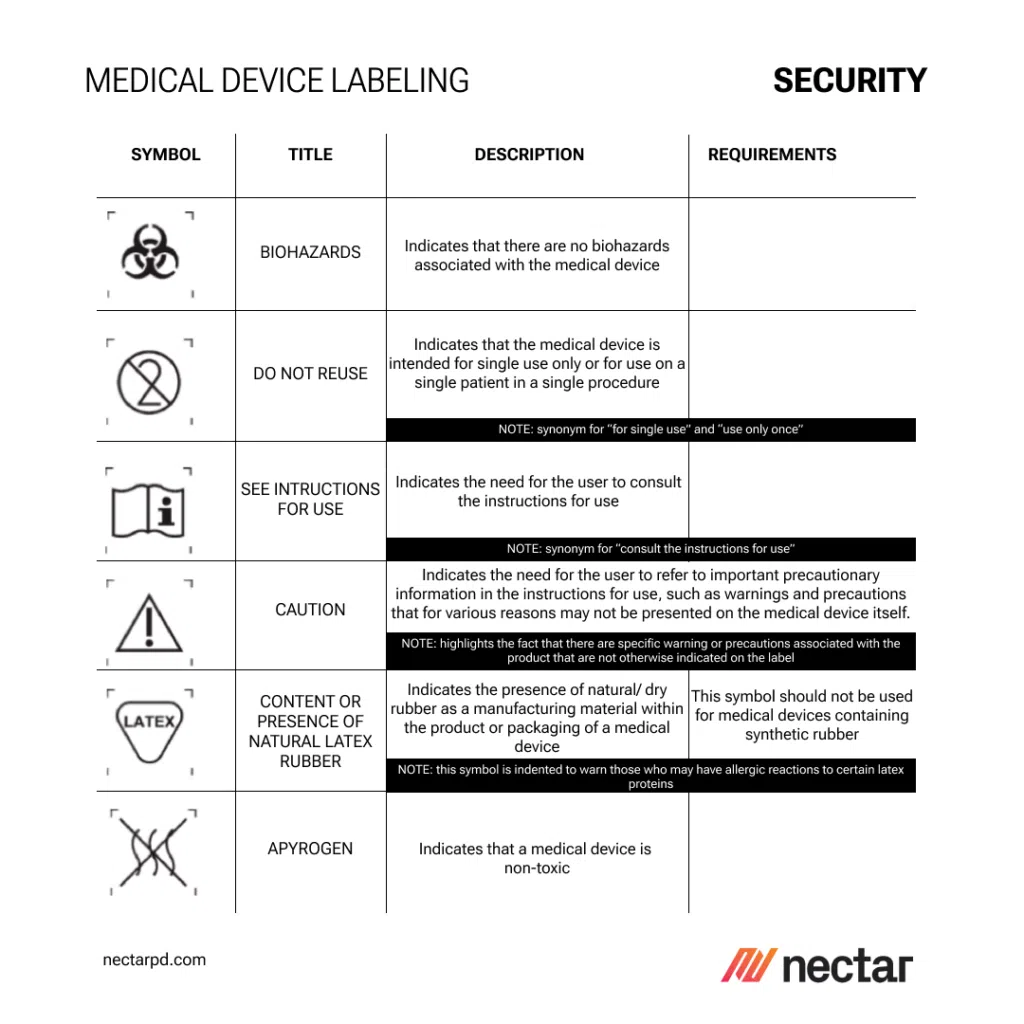

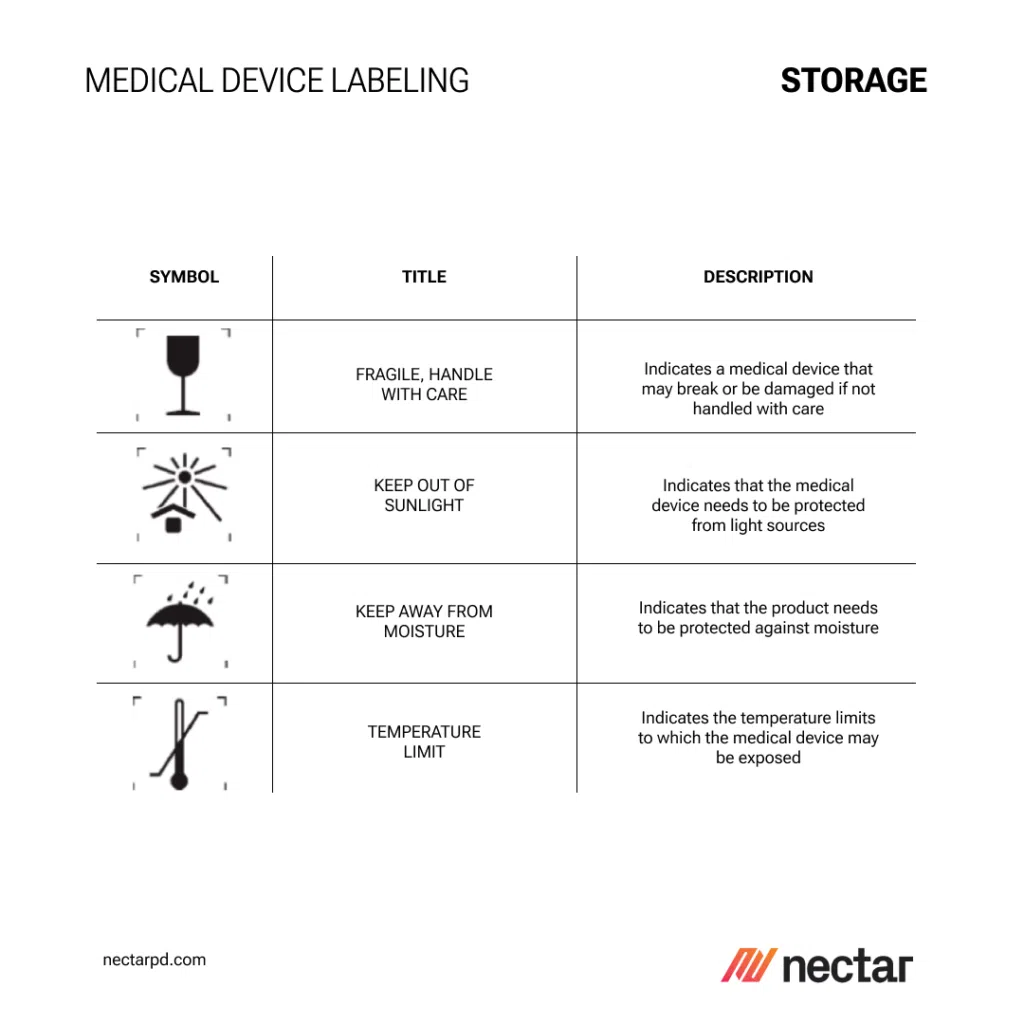
UDI, Traceability, and RFID: Advancing Beyond Basic Labeling in Medical Device Manufacturing
In the ever-evolving landscape of medical device manufacturing, Unique Device Identification (UDI), traceability, and Radio Frequency Identification (RFID) technology represent more than just advanced labeling techniques; they are pivotal elements in enhancing patient safety, supply chain efficiency, and regulatory compliance. This section explores how these technologies are revolutionizing the medical device industry by improving traceability and patient safety.
The Critical Role of Unique Device Identification (UDI)
UDI is a system implemented by regulatory bodies like the FDA to uniquely identify medical devices through their distribution and use. This system requires that each device bear a unique code, typically including a device identifier (DI) that specifies the model, and a production identifier (PI) providing production information. The UDI system enhances the ability to track devices, greatly improving recall effectiveness and surveillance of device-related adverse events. By incorporating UDIs in medical device labeling, manufacturers can greatly enhance the traceability and safety of their products.
Enhancing Traceability in the Supply Chain
Traceability in medical device manufacturing is not just a regulatory requirement; it is a critical component of patient safety and quality assurance. Effective traceability systems allow manufacturers to track the journey of a device from production to end-user, ensuring that any issues related to the device can be quickly and accurately addressed. This level of traceability is crucial in the event of recalls, helping to efficiently identify and rectify potential risks to patients.
Leveraging RFID Technology for Efficiency and Safety
RFID technology in medical device labeling enhances patient safety by providing each device with a unique digital identity that surpasses traditional barcodes. This advanced technology facilitates wireless tracking and inventory management, streamlining the supply chain and minimizing human error. RFID tags are capable of storing extensive data, such as manufacturing date, batch number, and expiration date. In clinical environments, the use of RFID technology is instrumental in managing medical device inventory, ensuring the correct device is administered to the appropriate patient, thereby bolstering patient safety.
RFID’s Role in Counterfeit Prevention and Brand Protection
Counterfeit medical devices threaten patient safety and compromise brand integrity. Employing RFID technology is an effective strategy against counterfeiting. Manufacturers can embed RFID tags into medical device labels to certify product authenticity. Scanning these tags provides instant verification of a device’s provenance, significantly obstructing the entry of counterfeit products into the market and protecting patient safety.
Integrating RFID with Medical Device Data Systems
The synergy between RFID technology and medical device data systems unlocks new horizons for data analysis and device management in the healthcare services sector. This powerful integration supports real-time tracking and monitoring, allowing manufacturers and healthcare providers to extract critical insights regarding device utilization, maintenance intervals, and patient outcomes. Such collaborative efforts can refine decision-making and contribute substantially to the enhancement of healthcare services.
UDI and RFID in Regulatory Compliance and 510(k) Submissions
In the realm of regulatory compliance and 510(k) submissions, the adoption of UDI and RFID technologies underscores a manufacturer’s dedication to safety and quality. These technologies are pivotal during the 510(k) submission process, illustrating a manufacturer’s approach to managing device traceability and reinforcing patient safety.
The Role of IFU and Patient Safety Information
Instructions for Use (IFU) and patient safety information are essential elements of medical device labeling, offering end-users comprehensive guidelines and risk/benefit data critical for risk management and the device’s safe usage. This section underscores the necessity for clarity and precision in IFUs, ensuring they are accessible and understandable to the intended audience. Nectar’s approach to usability testing for medical devices, an essential aspect of ensuring user-friendly and safe product designs.
Compliance with ISO 13485 and Risk Mitigation
ISO 13485 specifies the criteria for a quality management system tailored to the medical device industry, ensuring that organizations can consistently meet customer and regulatory requirements. Our multidisciplinary team of experts at Nectar Product Development is well-versed in these standards, aiding in mitigating risk and ensuring high-quality product development
Navigating 510(k) Submissions and Regulatory Pathways
The 510(k) submission process is a pivotal route for medical device manufacturers to validate that their products are as safe and effective as pre-existing devices. This section sheds light on maneuvering through this process, highlighting the importance of meticulous labeling in proving compliance and safeguarding user safety.
Innovating for Sustainability and Future Trends in Medical Device Labeling
As the medical device industry progresses, sustainability and innovation are becoming increasingly crucial, particularly in the area of medical device labeling. This evolution is driven by the need for environmentally responsible practices, advances in technology, and changing regulatory landscapes. In this section, we delve into how sustainability considerations and emerging trends are reshaping the future of medical device labeling.
Embracing Eco-Friendly Materials and Processes
Sustainability in medical device labeling isn’t just about the end product; it’s also about the journey there. Manufacturers are increasingly exploring eco-friendly materials for labels, such as biodegradable or recycled content, and adopting more energy-efficient printing methods. This shift not only reduces environmental impact but also resonates with a growing consumer base that values green practices.
Digital Labeling and E-Labels: A Technological Leap
The advent of digital labeling and electronic labels (e-labels) represents a significant technological shift in the industry. Digital labeling allows for greater flexibility in label design and content updates, which is particularly beneficial for manufacturers who deal with frequent regulatory changes or need to provide multilingual information. E-labels can be easily accessed via QR codes or NFC technology, reducing the need for extensive physical labeling and thereby contributing to waste reduction.
Leveraging RFID for Enhanced Efficiency and Traceability
RFID technology in medical device labeling isn’t just about compliance; it’s about stepping into a future of enhanced efficiency and traceability. RFID tags can store a wealth of information, from manufacturing details to expiration dates, and can be scanned without line-of-sight requirements. This technology not only streamlines supply chain management but also plays a vital role in ensuring the safety and authenticity of medical devices.
Tailoring Labeling to the Circular Economy
The concept of the circular economy is gaining traction in medical device labeling, encouraging manufacturers to think beyond the product’s immediate use. This approach involves designing labels and packaging that are reusable or part of a recycling stream, thus minimizing waste and supporting a more sustainable lifecycle for medical devices.
Balancing Regulatory Compliance and Sustainability
While innovating for sustainability, medical device manufacturers must balance their environmental objectives with stringent regulatory requirements. This balance requires a strategic approach to labeling, ensuring that all necessary information, such as UDI, manufacturer identification, and patient safety information, is provided in a sustainable yet compliant manner.
Future-Proofing with Smart Labels
Looking ahead, the integration of smart labels, which can change or relay information based on environmental factors or time, is a promising area. These innovations could offer dynamic ways to communicate with users, provide real-time updates, and even integrate with healthcare IT systems for better device management.
A Holistic Approach to Sustainable Labeling
In conclusion, innovating for sustainability in medical device labeling requires a holistic approach that encompasses environmental sustainability, technological advancements, regulatory compliance, and patient safety. As the industry moves forward, these innovations in medical device labeling practices are not only enhancing sustainability but also improving efficiency, traceability, and the user experience.
For an in-depth understanding of how sustainability is integrated into product development, explore Nectar Product Development’s expertise in sustainability and product development.scientific software and instrument development. Additionally, to stay updated with the latest trends and insights in product development, tune in to Nectar’s Podcast: The Product Development Book.
In conclusion, medical device labeling is a multifaceted process that requires careful consideration of regulatory standards, patient safety, and innovative practices. As we continue to develop innovative practices in medical device labeling, we must prioritize regulatory standards and patient safety. award-winning products in the medical device sector, it’s essential to prioritize comprehensive and compliant labeling strategies. For further inquiries or to discuss your medical device project with our team, feel free to Contact Us.























