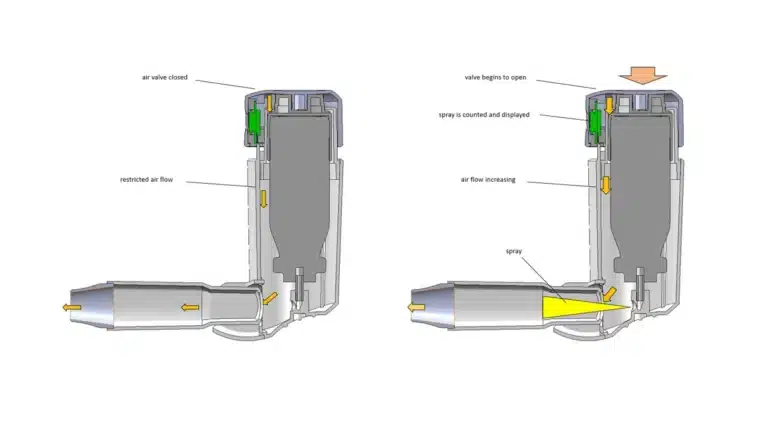
Breath Coordinated Inhaler
KOS Pharmaceuticals, a leader in the development of inhalation medication delivery technologies and now part of Abbott Labs, needed to develop a breath-coordinated inhaler that would administer asthma medication more precisely and effectively.

Delivering Inhalation Medications More Precisely
The pharmaceutical and medical device company turned to Nectar to create an inhalation system that would more efficiently deliver aerosol medication deep within the lungs during inspiration, with a minimum of medication loss either inside the mouth or as a result of system performance.
Measuring and Tracking Doses
Nectar undertook the challenge of creating a breath-coordinated inhaler that not only delivered medication in coordination with a patient’s breathing but also in a more precise and measured fashion. In addition to enabling the measurement and tracking of specific doses, the design had to include a valve for timing the release of medication in concert with inhalation, an air passage with a carefully engineered turbulent flow, and a compact, foldable shape to protect the unit from dirt and debris. All development also had to comply with federal Food and Drug Administration (FDA) requirements.
FDA-Compliant Development and Testing
The design that Nectar created had to both satisfy product goals and comply with FDA regulations governing development and testing. Nectar designed a metered-dosage system that tightly controlled dosage and counted the number of doses delivered. Development included all standard steps—including mechanical design, rapid prototyping, prototype tooling, part inspection, and assembly for clinical testing, as well as creation of a special testing apparatus for performance verification. The design was cost-efficient to manufacture and assemble in production quantities, and adapted to accommodate tolerance variations in standard medication canisters.